Table of Contents
- What is chemical recycling and what does it involve?
- Advanced pyrolysis vs. depolymerization
- What is advanced pyrolysis?
- What is depolymerization?
- What are the depolymerization routes?
- Strategic approach: when to use each technology?
- Impact of chemical recycling on Scope 3 emissions
- Conclusions
- References
- Frequently Asked Questions (FAQs)
Chemical recycling represents a technological evolution compared to the limits of traditional mechanical recycling. While the latter preserves the polymer structure and may lose properties after multiple cycles, chemical recycling acts at the molecular level, breaking polymer chains to recover monomers, hydrocarbons, or reusable chemical feedstocks. This approach allows the treatment of complex, contaminated, or multilayer waste that could hardly be reincorporated using conventional methods.
Within the framework of the circular economy, advanced recycling seeks to close the materials loop not only through physical reuse but through molecular reconstruction. The most relevant technologies in this field are advanced pyrolysis and various depolymerization routes, each with specific applications depending on the type of polymer and industrial objective.
What is chemical recycling and what does it involve?
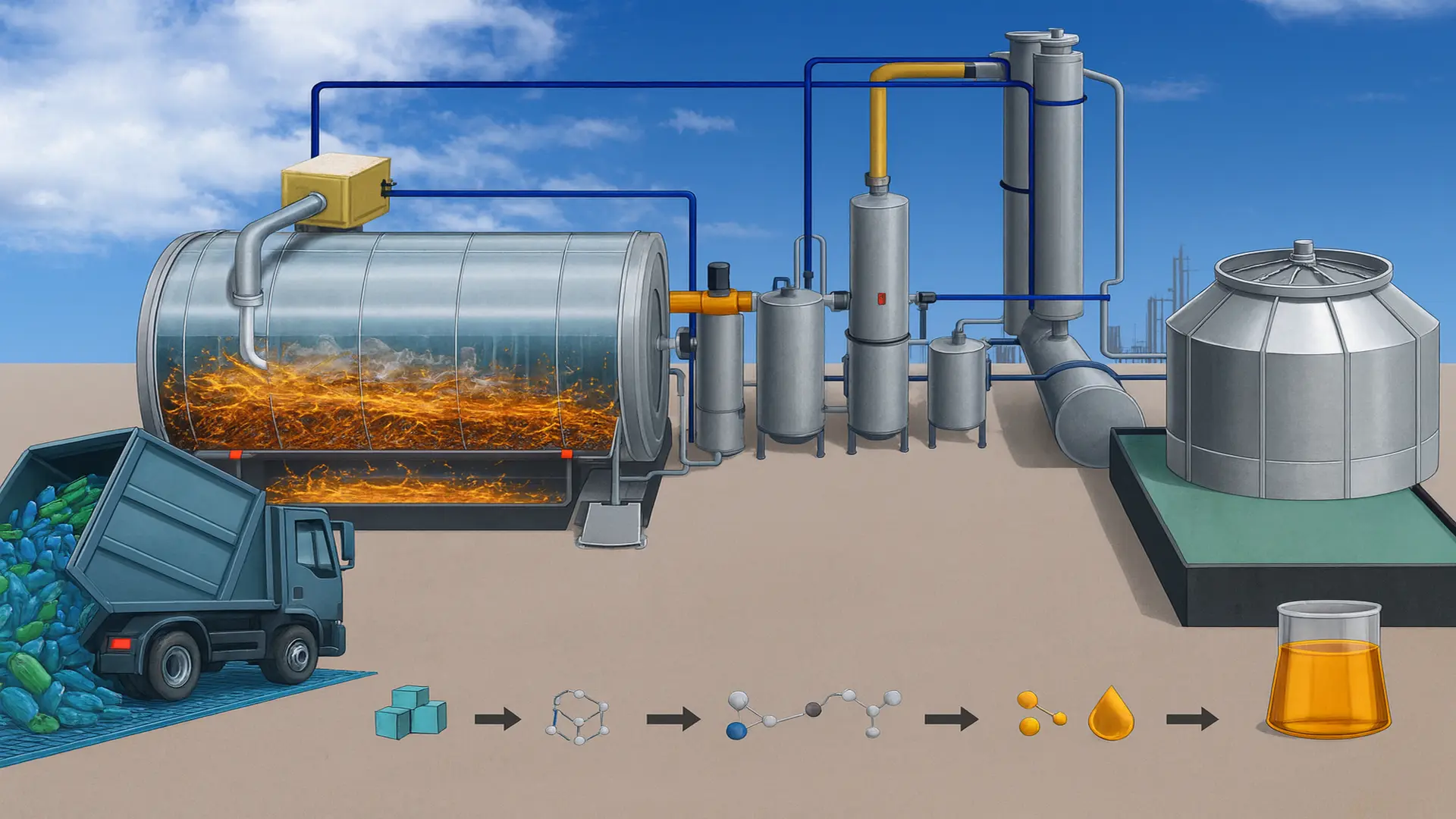
Chemical recycling has become one of the strategic pillars of the transition towards a truly circular economy. It consists of a set of industrial processes that transform plastic waste at the molecular level to convert it again into useful chemical feedstocks.
Unlike mechanical recycling, which preserves the polymer structure but may degrade its properties after multiple cycles, chemical recycling transforms plastic waste at the molecular level. This means that polymer chains are broken to recover monomers, hydrocarbon fractions, or feedstocks that can be reincorporated into the petrochemical industry with quality equivalent to virgin material.
In this context, technologies such as advanced pyrolysis and various depolymerization routes constitute the core of so-called advanced recycling. Simply put, it involves “disassembling” the plastic to recover its basic components or convert it into new molecules that can be used to manufacture plastics of equivalent quality to virgin material.
How does chemical recycling work?
The process can vary depending on the technology used, but generally includes three main stages:
First, the plastic waste is sorted and conditioned. Then it undergoes a chemical or thermochemical process that breaks the molecular chains. Finally, the obtained products are purified and reincorporated as feedstock in petrochemical or polymerization processes.
Advanced pyrolysis vs. depolymerization
The chemical valorization of plastic waste has become a strategic point within the circular economy. However, not all thermal and chemical processes are equivalent. Two of the most discussed technologies today are advanced pyrolysis and depolymerization—terms often used interchangeably but representing distinct technical approaches.
It is important to understand the difference between these two processes to select the appropriate technology depending on the polymer type, required purity level, and target market of the final product.
What is advanced pyrolysis?
Advanced pyrolysis is a thermochemical process that decomposes organic materials in the absence of oxygen at high temperatures (350–700 °C). In the case of plastic waste, the main output is pyrolysis oil, along with gas and solid char.
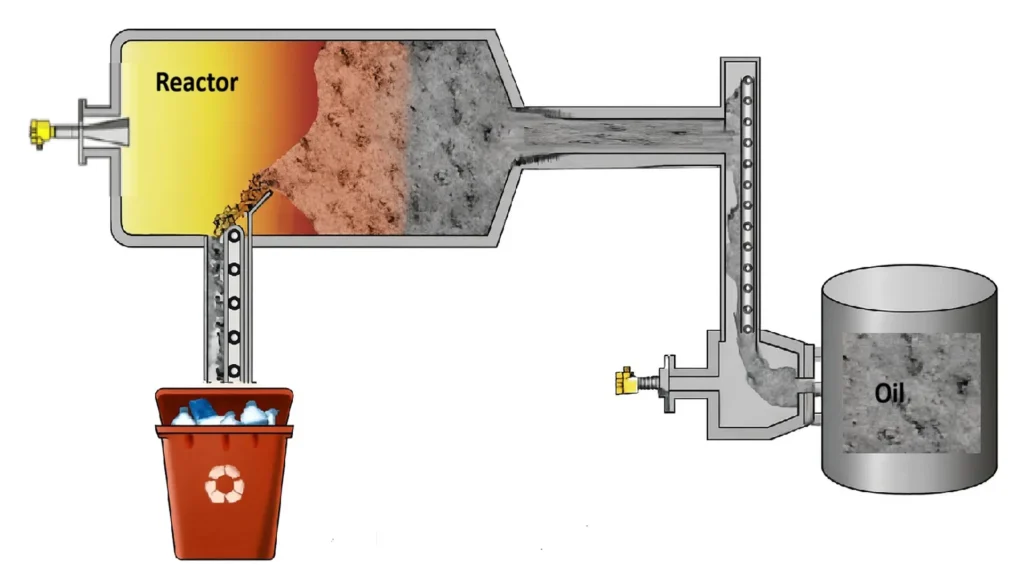
Key features
- Non-selective at the molecular level.
- Mainly applied to addition polymers such as:
- Polyethylene (PE)
- Polypropylene (PP)
- Polystyrene (PS)
- Produces complex hydrocarbon mixtures.
- Requires further refining to obtain petrochemical-grade products.
Advanced pyrolysis is optimized through
- Fluidized bed reactors
- Catalytic systems
- Thermal residence control
- Improved energy recovery
Its goal is not to reconstruct the original monomer but to generate feedstocks equivalent to fuels or petrochemical naphtha.
What is depolymerization?
Depolymerization is a selective chemical process that breaks polymer bonds to recover original monomers or high-value chemical intermediates.
It is mainly applied to condensation polymers, such as:
- PET (polyethylene terephthalate)
- Polyamides (nylon)
- Polyurethanes
- Polycarbonates
Unlike pyrolysis, depolymerization aims to:
- Recover molecular purity
- Reintroduce the monomer into the production chain
- Enable “closed-loop” chemical recycling
Technical differences between Advanced Pyrolysis and Depolymerization
| Criteria | Advanced Pyrolysis | Depolymerization |
|---|---|---|
| Process type | Non-selective thermochemical | Selective chemical |
| Temperature | High (350–700 °C) | Moderate (150–300 °C in many cases) |
| Oxygen | Completely absent | May include solvents/reactants |
| Polymer type | Mainly addition (PE, PP) | Mainly condensation (PET, nylon) |
| Main product | Hydrocarbon mixture | Defined monomers or oligomers |
| Product purity | Requires refining | High molecular selectivity |
| Petrochemical integration | Upcycling via cracking | Direct closed-loop recycling |
What are the depolymerization routes?
Depolymerization routes depend on both the chemical nature of the polymer and the reactive agent used to induce bond cleavage. Generally, they are mainly applied to condensation polymers, whose ester, amide, or carbonate bonds can be selectively broken to recover monomers or high-value intermediates.
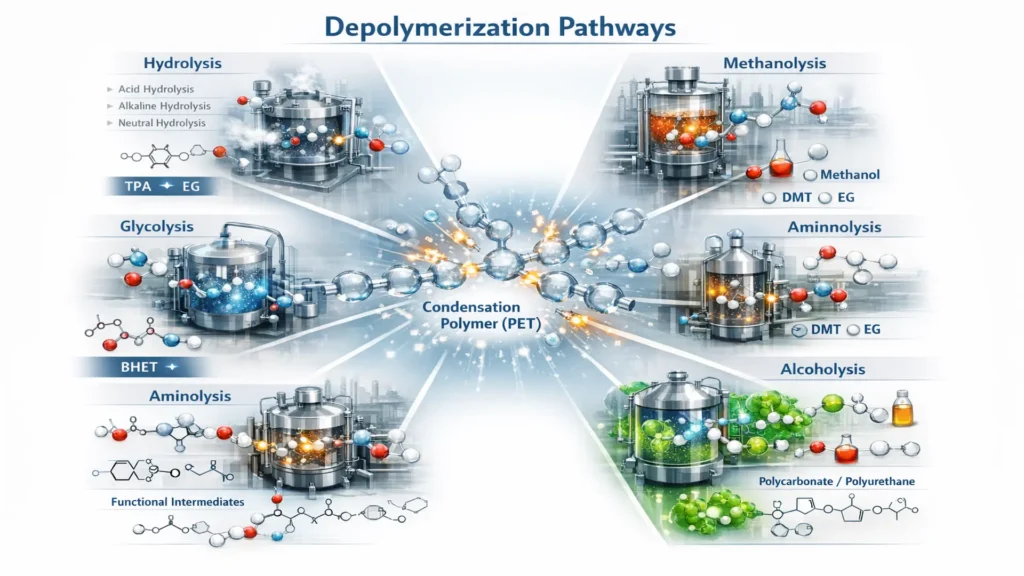
Below are the main routes used industrially and in technological development:
- Hydrolysis: Hydrolysis involves breaking the polymer using water, typically under high temperature and pressure conditions that favor ester or amide bond cleavage. Its typical application is in PET recycling, where the process recovers terephthalic acid (TPA) and ethylene glycol (EG), the two original monomers. Depending on the reaction medium, it can be classified as acidic hydrolysis (using acid solutions), alkaline hydrolysis (using strong bases), or neutral hydrolysis (operating with water alone under more severe conditions). Each variant differs in terms of corrosivity, energy consumption, and subsequent purification steps.
- Glycolysis: Glycolysis involves reacting the polymer with a glycol, commonly ethylene glycol, promoting ester bond cleavage and the formation of oligomers or functionalized monomers. For PET, the main product is bis(2-hydroxyethyl) terephthalate (BHET), which can be purified and reused in new polymer synthesis. This route is one of the most industrially developed due to its relative energy efficiency, moderate operating conditions, and ease of integration into semi-closed chemical recycling schemes.
- Methanolysis: Methanolysis is based on reacting the polymer with methanol, producing mainly dimethyl terephthalate (DMT) and ethylene glycol for PET. This process was widely used in the petrochemical industry before the consolidation of direct production from purified terephthalic acid. Methanolysis yields high-purity products but requires controlled pressure and temperature conditions and methanol recovery systems to maintain economic viability.
- Aminolysis: Aminolysis uses amines as reactive agents to break ester or amide bonds in certain polymers. It is applied to both PET and polyurethanes, generating functional compounds that can serve as intermediates for resins, adhesives, or other polymeric materials. This route allows the introduction of specific functional groups in the final product, expanding its application in specialized formulations.
- Enzymatic depolymerization: Enzymatic depolymerization uses specific enzymes capable of selectively attacking polymer bonds, particularly in PET. It is an emerging technology that operates at much lower temperatures than conventional thermochemical processes and offers high molecular selectivity. Advantages include lower energy consumption and potential carbon footprint reduction. However, it still faces significant limitations related to industrial scalability and enzyme production cost and stability.
- Alcoholysis: Alcoholysis involves reacting the polymer with alcohols other than methanol or glycols, enabling controlled bond cleavage in materials such as polycarbonates and polyurethanes. Depending on the alcohol used, different chemical intermediates can be obtained and later reincorporated into synthesis processes. This route offers chemical flexibility and can adapt to specific valorization schemes depending on the type of waste treated.
Strategic approach: when to use each technology?
From a strategic perspective, the choice between advanced pyrolysis and depolymerization fundamentally depends on the composition of the waste stream and the final product objective. For mixed, heterogeneous, or highly contaminated plastic streams, advanced pyrolysis is generally more suitable due to its robustness and impurity tolerance. Conversely, for relatively pure, single-material streams, such as source-separated PET bottles, depolymerization allows recovery of high-purity monomers and enables closed-loop molecular recycling.
If the goal is to produce fuels or generate feedstock equivalent to petrochemical streams for refinery integration, pyrolysis is a viable option. On the other hand, if the aim is to maintain the polymer’s molecular value and reincorporate it directly into the production cycle with minimal quality loss, depolymerization is the most technologically coherent alternative within an advanced circular economy model.
Impact of chemical recycling on Scope 3 emissions
Reducing indirect emissions along the value chain has become one of the greatest challenges for the petrochemical and materials industries. According to the Greenhouse Gas Protocol, Scope 3 emissions include those generated outside a company’s direct operational boundaries but linked to its activities, such as raw material production, transportation, processing, and product end-of-life. In this context, chemical recycling emerges as a strategic tool with structural impact on corporate carbon footprint.
Substitution of fossil feedstock
One of the main contributions of chemical recycling is the partial replacement of virgin fossil feedstocks with secondary feedstocks recovered from plastic waste. In processes such as advanced pyrolysis, mixed waste can be transformed into oils equivalent to petrochemical naphtha, while depolymerization recovers high-purity monomers that can be directly reincorporated into new polymer synthesis.
This substitution reduces the demand for extraction and processing of fossil resources, activities that account for a significant portion of upstream emissions. From the perspective of resin manufacturers and consumer brands, integrating chemically recycled content reduces the carbon intensity associated with the “purchased goods and services” category, one of the most relevant within Scope 3. Thus, chemical recycling not only manages waste but also acts as a mechanism for partially decoupling production growth from virgin resource consumption.
Reduction of end-of-life emissions
Plastic end-of-life represents another relevant source of indirect emissions, especially when waste is incinerated without efficient energy recovery or landfilled, generating degradation- and transport-related emissions. Chemical recycling offers an alternative that reintegrates the carbon contained in plastics into the production cycle, avoiding direct CO₂ release or permanent loss from the economic system.
By converting waste into new feedstocks, the fraction destined for disposal is reduced, and the value of carbon originally incorporated into the polymer is extended. This approach helps reduce reported emissions in categories related to waste treatment and end-of-life products. Systemically, it advances a molecular circularity model where carbon remains in use as long as possible, improving material and climate efficiency.
Reconfiguration of the petrochemical value chain
Beyond direct impacts on feedstocks and end-of-life, chemical recycling drives structural transformation in the petrochemical value chain. The integration of pyrolysis or depolymerization units into industrial complexes creates circular carbon flows coexisting with traditional fossil streams. This technological hybridization shifts the linear logic of extraction–production–waste towards more circular and resilient schemes.
Additionally, traceability through mass balance schemes and recycled content certifications strengthens supply chain transparency, facilitating more robust ESG reporting and decarbonization strategies aligned with international climate commitments. In this scenario, chemical recycling becomes a structural component of the transition toward low-carbon petrochemicals, where Scope 3 emission management is addressed through redesign of the production system itself.
Conclusions
Advanced pyrolysis and depolymerization represent complementary technological approaches within plastic chemical recycling but respond to different valorization logics. While pyrolysis prioritizes thermochemical conversion of heterogeneous streams into reusable hydrocarbon fractions as petrochemical feedstock, depolymerization seeks to preserve the polymer’s molecular value through selective recovery of high-purity monomers or intermediates.
The technical and economic feasibility of each route mainly depends on waste composition, degree of prior separation, and target market of the final product. For single-material streams such as post-consumer PET, hydrolysis, glycolysis, or methanolysis enable closed-loop recycling with minimal quality loss. In contrast, for mixed or contaminated waste, advanced pyrolysis offers greater operational flexibility and industrial robustness.
From a strategic circular economy perspective, integrating both technologies within the same industrial ecosystem maximizes value recovery and reduces dependence on virgin feedstock. The global trend points toward hybrid systems where mechanical separation, selective depolymerization, and advanced pyrolysis operate complementarily to optimize material, energy, and environmental efficiency.
References
- Al-Sabagh, A. M., Yehia, F. Z., Eshaq, G., Rabie, A. M., & ElMetwally, A. E. (2016). Greener routes for recycling of polyethylene terephthalate. Egyptian Journal of Petroleum, 25(1), 53–64. https://doi.org/10.1016/j.ejpe.2015.03.001
- Rahimi, A., & García, J. M. (2017). Chemical recycling of waste plastics for new materials production. Nature Reviews Chemistry, 1(6), 0046. https://doi.org/10.1038/s41570-017-0046
- Ragaert, K., Delva, L., & Van Geem, K. (2017). Mechanical and chemical recycling of solid plastic waste. Waste Management, 69, 24–58. https://doi.org/10.1016/j.wasman.2017.07.044
Frequently Asked Questions (FAQs)
Which plastics are suitable for advanced pyrolysis today?
In current industrial practice, plastics most suitable for advanced pyrolysis are polyethylene, polypropylene, and polystyrene. These materials have hydrocarbon chains that efficiently convert into usable liquid fractions. PVC requires pretreatment to remove chlorine, and PET is generally better processed via depolymerization due to its chemical structure.
When is PET depolymerization recommended in industry?
PET depolymerization is recommended when mechanically recycled material does not meet required specifications, when the waste is contaminated, or when producing resin suitable for demanding applications such as food packaging. Recovering original monomers allows restarting the production cycle with purity standards comparable to virgin material.
What are examples of chemical recycling?
Examples of chemical recycling include plastic pyrolysis to produce refinery feedstock, PET glycolysis to recover monomers, methanolysis of polyester materials, depolymerization of nylon into caprolactam, and gasification of mixed plastic waste into synthesis gas. These processes convert polymer waste into valuable chemical intermediates rather than simply melting and reshaping the material.
What is the main disadvantage of chemical recycling?
The main disadvantage of chemical recycling is its high energy demand and capital investment requirement. The technology involves complex reactors, strict operational controls, and stable feedstock supply. Additionally, regulatory debates continue in some regions regarding whether certain outputs, particularly fuels derived from pyrolysis, should be classified strictly as recycling or as energy recovery.