By : Dr. Franyi Sarmiento, Ph.D., Inspenet, March 11, 2022
Researchers at the University of California Santa Cruz (UCSC, USA) demonstrated that an easily produced compound of gallium and aluminum creates aluminum nanoparticles that react rapidly with water at room temperature to produce large amounts of hydrogen, without the need for having no power input.
“I’ve never seen anything like it,” Scott Oliver, a UCSC professor of chemistry, writes in an article posted on the University website. “We don’t need any energy input, and hydrogen bubbles like crazy,” adds the researcher, who shares the direction of this work with Bakthan Singaram, professor of chemistry and biochemistry, published in the February issue of the Applied Nano Materials journal.
Aluminum is a highly reactive metal that can extract oxygen from water molecules to generate hydrogen gas. Its widespread use in products that get wet does not present any danger because aluminum reacts instantly with air to acquire a layer of aluminum oxide, which blocks further reactions.
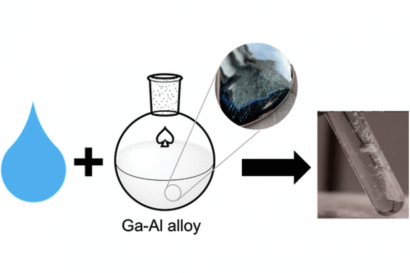
The reaction of aluminum and gallium with water has been known since the 1970s. It works because gallium, which remains liquid over a very wide temperature range, removes the passive layer of aluminum oxide, allowing direct contact of aluminum with water. This new research, however, includes several innovations and findings that could lead to practical applications.
Using scanning electron microscopy and X-ray diffraction, the researchers showed the formation of aluminum nanoparticles in a 3:1 gallium-aluminum compound, which they found to be the optimal ratio for hydrogen production.
In this gallium-rich compound, the gallium serves both to dissolve the aluminum oxide coating and to separate the aluminum into nanoparticles. “The gallium separates the nanoparticles and prevents them from aggregating into larger particles,” says Singaram. “People have struggled to make aluminum nanoparticles, and here we are making them under normal atmospheric pressure and room temperature conditions.”
Making the compound requires nothing more than simple hand mixing. “Our method uses a small amount of aluminum, which ensures that most of the gallium dissolves as discrete nanoparticles,” says the researcher. “This generates a much higher, almost complete amount of hydrogen compared to the theoretical value based on the amount of aluminum. It also makes it easier to recover gallium for reuse.”
The composite material can also be made from readily available aluminum, including used sheets or cans, and can then be stored for long periods, covered with cyclohexane to protect from moisture.
As for gallium, although it is not abundant and is relatively expensive, it can be recovered and reused several times without losing effectiveness, explains Singaram. Mind you, it remains to be seen if this process can be scaled up to make it practical for commercial-scale hydrogen production.
Source and photo : https://www.energias-renovables.com/hidrogeno/cientificos-obtiienen-hidrogeno-de-manera-eficiente-a-20220223